Targeted Nutritional support™
Procite-DTM
Catecholamine Formula*
Procite-D™ is a catecholamine support formula specifically targeting dopamine while also supporting PEA, norepinephrine, epinephrine and beta-endorphin pathways.* Cofactors and/or methylated B vitamins critical for synthesis are included along with the proprietary Neuro Support Blend™ (NSB) for enhanced absorption.*
Encapsulating the Art & Science of Functional nutrition™
Formula Overview
Use Procite-D™ to support the rebalancing process targeting dopamine/catecholamines.*
Mucuna pruriens (standardized to 20% L-DOPA) Natural source of L-DOPA, a direct precursor to the catecholamines dopamine, norepinephrine and epinephrine.*
DL-Phenylalanine DL-phenylalanine acts a precursor to PEA as well as the catecholamine pathway.*
N-Acetyl-L-Tyrosine Precursor to L-DOPA, the direct precursor to the catecholamines dopamine, norepinephrine and epinephrine.*
N-Acetyl-L-Cysteine Antioxidant.* Helps maintain cell health during catecholamine supplementation.*1
Folate (as (6S)-5-methyl-tetrahydro folic acid, glucosamine salt) (Quatrefolic®) Cofactor in methylation reactions essential for the synthesis of dopamine, norepinephrine and epinephrine.*
Selenium Supports selenoproteins, important antioxidant enzymes.* Data shows reduced selenium activity can impact healthy dopamine, reducing activity. 2
Vitamin B6 (as pyridoxal-5’-phosphate) Active form of B6. Cofactor in the conversion of L-DOPA to dopamine.*
Vitamin B12 (as Methylcobalamin) Coenzyme form of Vitamin B12.* Methyl donor.*
NeuroSupport Blend™ (NSB) Proprietary mixture composed of Bioperine® and plant-based enzymes designed to maximize nutrient absorption.*
Serving Size: 1 Capsule
Servings Per Container: 60 Capsules
Procite-D™ Formula 435.75 mg
Vitamin B6 (as pyridoxal-5-phosphate)
Folate (as (6S)-5-methyl-tetrahydro folic acid, glucosamine salt) (Quatrefolic®)
Vitamin B12 (as methylcobalamin)
Selenium (as L-selenomethionine)
N-Acetyl-L-Tyrosine
N-Acetyl-L-Cysteine
DL-Phenylalanine
Mucuna pruriens (std. to 20% l-Dopa) (seed)
NSB™ Proprietary Blend 102.5 mg
BioPerine® (Black Pepper Extract) (standardized to 95% piperine)(Piper nigrum) (fruit)
Protease
Amylase
Lipase

NSB™ is a trademark of Sanesco International Inc.
![]()
Quatrefolic® is a registered trademark of Gnosis S.p.A, U.S. Patent No. 7,947,662
![]()
BioPerine® is a registered trademark and a product of Sabinsa Corp.
Dopamine balance is important to maintain:
- Motivation3,4
- Balanced impulse control5,6
- Cravings, healthy pleasure/reward feelings7-10
- Healthy libido11-14
Norepinephrine balance is important to maintain:
- Healthy stress response15-17
- Healthy mood
- Temperature balance18-20
- Balanced energy21
- Healthy focus and cognitive function22,23
- Healthy bodily comfort24,25
Epinephrine balance is important to maintain:
- Healthy stress response15,16,27
- Balanced emotional health28
- Healthy blood sugar maintenance16,29
- Weight management balance27,31,32
PEA balance is important to maintain:
- Mental and emotional health balance32,33
- Healthy focus33, 34
- Noh J, Kim E, Kang J, et al. Experimental Neurology. 159, 217–224.
- Gashu, D and Stoecker BJ. Selenium and Cognition: Mechanism and Evidence. In: Preedy V., Patel V. (eds) Handbook of Famine, Starvation, and Nutrient Deprivation. Springer, Cham 2017:1-17. Accessed on August 6, 2019 https://link.springer.com/referenceworkentry/10.1007%2F978-3-319-40007-5_21-2
- Palmiter RD. Ann N Y Acad Sci. 2008;1129:35-46.
- Felger JC and Treadway MT. Neuropsychopharmacology. 2017 Jan;42(1):216-241.
- Kelley BJ, et al. Parkinsons Dis. 2012;2012:603631.
- Athanasoulia-Kaspar AP, et al. Endocr Connect. 2018 Feb;7(2):R88-R94.
- Blum K, et al. Curr Neuropharmacol. 2017;15(1):184-194.
- Burger KS and Stice E. Am J Clin Nutr. 2012 Apr;95(4):810-7.
- Stice E, et al. J Neurosci. 2010 Sep 29;30(39):13105-9.
- Lennerz B and Lennerz JK. Clin Chem. 2018 Jan;64(1):64-71.
- Abler B, et al. Neuropsychopharmacology. 2011 Aug;36(9):1837-47.
- Goldstein I, et al. Mao Clin Proc. 2017 Jan;92(1):114-128.
- Just MJ. Neuropsychiatr. Dis Treat. 2015 Jul 8;11:1655-61.
- Stahl SM. J Clin Psychiatry. 2010 Jul;71(7):821-2.
- Lieberman HR, et al. Physiol Behav. 2016 Oct 15;165:86-97.
- Wong H, et al. Cureus. 2019 Aug 24;11(8):e5474.
- Flak JN, et al. Eur J NeuroSci. 2014 Jun;39(11):1903-11.
- Bansal R and Aggarwal N. J Midlife Health. 2019 Jan-Mar;10(1):6-13.
- Freedman RR. Fertil Steril. 1998 Aug;70(2):332-7.
- Gordon JL, et al. Menopause. 2016 Nov;23(11):1189-1198.
- Brennan AR and Arnsten AF. Ann N Y Acad Sci. 2008;1129:236-45.
- Arnsten AF and Pliszka SR. Pharmacol Biochem Behav. 2011 Aug;99(2):211-6.
- Borodovitsyna O, et al. Neural Plast. 2017;2017:6031478.
- Ho KY, et al. Br J Anaesth. 2010 Sep;105(3):371-6.
- Hoshino H, et al. Anesth Analg. 2015 Feb;120(2):460-6.
- Lee ZS, et al. Metabolism. 2001 Feb;50(2):135-43.
- Rief W, et al. Psychosom Med. 2010 Oct;72(8):755-62.
- Paine NJ, et al. Psychosom Med. 2015 Feb-Mar;77(2):136-44.
- Howlett K, et al. J Physiol. 1999 Sep 15;519 Pt 3:911-21.
- Corbi GM, et al. J Clin Endocrinol Metab. 2002 May;87(5):2080-3.
- Couillard C, et al. Obes. Res. 2002 Jan;10(1):6-13.
- Szabo A, et al. Br J Sports Med. 2001 Oct;35(5):342-3.
- Kaur N and Kumari B. World J Pharm & Pharmaceut Sci. 2016 Mar;5(4):743-750.
- Irsfeld M, et al. Webmedcentral. 2013 Sep 30;4(9). pii: 4409.

targeted Dopamine nutrition
Featured Ingredients
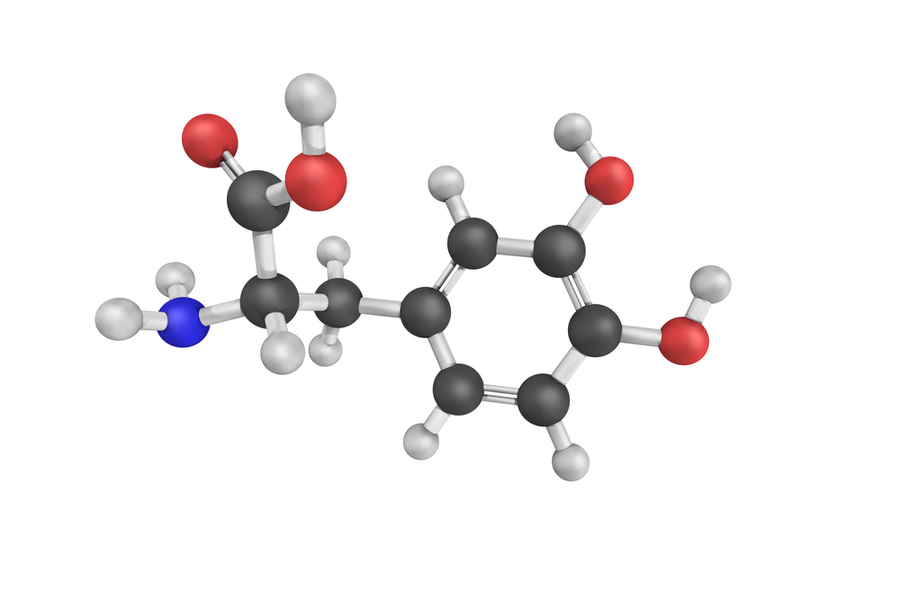
DL-phenylalanine
Amino Acid
DL-phenylalanine acts a precursor to PEA as well as the catecholamine pathway*

Mucuna pruriens
Herbal Ingredient
Natural source of L-DOPA, direct precursor to the catecholamines dopamine, norepinephrine and epinephrine.*
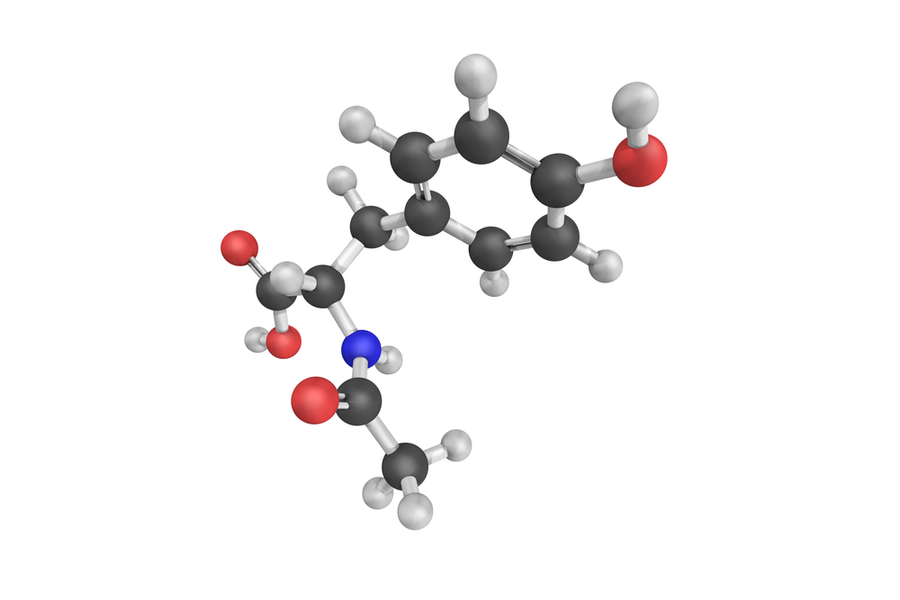
N-acetyl-L-tyrosine
Amino Acid
N-Acetyl-L-Tyrosine Precursor to L-DOPA, the direct precursor to the catecholamines dopamine, norepinephrine and epinephrine.*