Targeted Nutritional support™
Methyl FMTTM
Integrated Support
Fundamental methylation support and complete cofactor fuel designed for the entire family, from genetics and mood to healthy homocysteine*
Encapsulating the Art & Science of Functional nutrition™
Formula Overview
Fundamental methylation support with complete cofactor fuel to address the entire folate, methylation, and transulferation pathways, with suggested use starting at age 4.* Designed as a gentle, well-rounded blend for those looking to slowly start broad methylation and homocysteine support with the flexibility to easily scale as needed.*
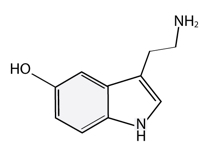
Methyl FMT combines activated forms of B6, folate, and B12 with B2, B3, trimethylglycine (betaine) and cofactors magnesium and zinc. With these ingredients supporting the folate cycle (in turn biopterin) and methylation pathways, the production of serotonin, dopamine, norepinephrine, epinephrine, and melatonin are also promoted. These neurotransmitters and hormone consequently impact mood, cravings, cognition, temperature regulation, immune activity, energy, motivation, and the sleep-wake cycle.*
Trusted by healthcare providers for over 20 years with its specialized neurotransmitter supporting formulas, NeuroScience is a recognized brain health leader for designing impactful, targeted dietary supplement blends that promote sleep, stress, mood, energy, and cognitive health.
Riboflavin (B2)
- Essential for MTHFR and MTRR coenzyme FAD in the folate cycle
- Riboflavin supplementation has been shown to significantly reduce homocysteine levels in those with genetic predisposition*
Niacin (B3)
- Niacin, in the form of NAD, is a necessary cofactor for the enzymes DHFR in the folate/tetrahydro- biopterin cycles and S-adenosylhomocysteine hydrolase in the methionine cycle
Vitamin B6
- Essential cofactor for multiple enzymes in the methylation and transsulfuration pathways including SHMT, CBS, and CTH
- Essential cofactor for synthesis of neurotransmitters such as dopamine, GABA, norepinephrine, epinephrine, and serotonin
L-5-MTHF
- Primary form of folate used by the body; does not require metabolism for absorption
- Patented crystallization form for preferred stability*
Vitamin B12
- Essential cofactor for the metabolism of L-5-MTHF in the folate pathway and homocysteine activity in the methionine pathway
Magnesium
- Specific form of magnesium bound to glycine molecules naturally creating a low weight chelate with enhanced bioavailability*
- Required cofactor for methionine adenosyl- transferase, the enzyme responsible for synthesizing SAMe in the methionine pathway
Zinc
- Essential cofactor enzymes in the methylation pathway including MTR and BHMT
Trimethylglycine
- Potent methyl donor and substrate for BHMT, sparing choline resources*
- Trimethylglycine supplementation has been shown to support healthy homocysteine levels*
Serving Size: 2 Capsules
Servings Per Container: 30
Methyl FMT Formula:
- Riboflavin (as riboflavin 5′-phosphate)
- Niacin (as niacinamide)
- Vitamin B6 (as pyridoxal 5’phosphate)
- Folate (as L-5-methyltetrahydrofolate, calcium salt)**
- Vitamin B12 (as methylcobalamin)
- Magnesium (as magnesium bisglycinate chelate)***
- Zinc (as zinc bisglycinate chelate)***
- Trimethylglycine (as betaine anhydrous)
**Magnafolate is a registered trademark of Lianyungang Jinkang Pharmaceutical Technology Co., Ltd.
***ALBION MINERALS Trademark of ALBION LABORATORIES, INC.
Formulated vegan, non-GMO, and free of eggs, fish, gluten, milk, peanuts, shellfish, soy, and tree nuts. All lots of NeuroScience products undergo independent, third-party testing.
Symptoms:
- Adrenal
- Anxiousness/Stress
- Cognition/Focused Attention
- Energy
- Mood
- Sleep
- Weight Management
Patient Profile:
- Methylation and enzyme support