lab testing Partners
Lab partners are leading specialists in the research, analysis, and development of biomarkers associated with hypothalamic-pituitary-adrenal (HPA) axis function. Optimal HPA function is foundational to peak performance of the body’s neuroendocrine communication system.
NON-Diagnostic neuroendocrine testing
Specialized Laboratory Testing for Neurotransmitters and Hormones
BIOMARKERS TESTED
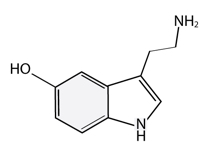
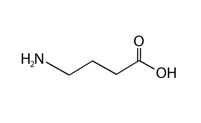
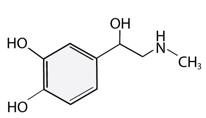
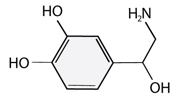
Neurotransmitters
Adrenal Hormones
Sex Hormones
COLA/CLIA Accredited
Lab Partners Spotlight
Our lab partners employ the gold standard in urinary neurotransmitter testing. UHPLC Triple Quadrupole Mass Spectrometry (MS) technology offers the highest level of sensitivity and specificity for measuring neurotransmitter activity. Salivary hormones are measured using ELISA. This technology provides highly accurate and reproducible results to ensure healthcare providers have the lab numbers and data to help to make effective clinical decisions.
Our lab partners have an extensive quality control program, including:
- 3rd party proficiency testing with External Quality Assurance Services® (EQAS), The College of American Pathology (CAP), and the American Proficiency Institute
- Blind samples testing with other labs and internally
- 2 levels of quality controls required with every test performed and on every day of testing
- QC values are analyzed using Levy Jennings charts and trends monitored
- Biannual COLA inspections verifying SOPs and guideline compliance
- Annual competency assessments of lab employees
- Quarterly self-inspections

The proprietary Laboratory Information Management System (LIMS) offers convenient, real-time access to patient results 24/7. NeuroLIMS is HIPAA secure and protects the patient’s personal health information while allowing healthcare providers easy access to test results.

trusted & reliable laboratory partnerships
Accreditation and Certifications
Our lab partners are both CLIA certified and COLA accredited. COLA Inc. is a physician-directed organization whose purpose is to promote excellence in laboratory medicine and patient care through a program of voluntary education, consultation, and accreditation.
The organization is an independent, non-profit accreditor whose education-based program and standards enable clinical laboratories and staff to meet U.S. CLIA 88 and other regulatory requirements.