Phenylethylamine (PEA), also known as beta-phenylethylamine or 2-phenylethylamine, is a central nervous system stimulant. Normal amounts of PEA produced in the body support healthy energy levels as well as attention and focus, well-being, happiness, and cognitive function.1
PEA is a biogenic monoamine, or a substance with one amino acid group connected to an aromatic ring that acts as a neurotransmitter and/or hormone in the body. However, PEA is found at very low levels in the central nervous system and is therefore called a trace amine. PEA does not act like a classic neurotransmitter,1 and its molecular mechanism of action differs from other biogenic amines, such as serotonin or dopamine.1
What is PEA and How Does It Work?
PEA, as a trace amine, is an endogenous amphetamine, and present in only small amounts in the brain compared to classic neurotransmitter amines such as norepinephrine, dopamine, and serotonin.1 PEA is a neuromodulator, influencing the effects of central monoamine neurotransmitters. Trace amines like PEA are synthesized in monoaminergic nerve terminals and have their own set of receptors called trace amine-associated receptors (TAARs). These receptors are widely expressed in the brain and are involved in:2
- Attention
- Cravings
- Focus
- Mental health
- Mood
- Nerve health
How Is Phenylethylamine Made and Where Is It Found in the Body?
PEA is made in monoaminergic neurons located in the central nervous system from the amino acid L-phenylalanine. This is accomplished through the enzyme amino acid decarboxylase (AADC), which converts L-phenylalanine to PEA at a rate similar to the central synthesis of dopamine.3 Unlike classical neurotransmitters, PEA is not stored in neurons, but rather is broken down primarily through monoamine oxidase (MAO-B), which catabolizes PEA at a very rapid rate to phenylacetic acid (PAA), which will be discussed further.4 Because it is broken down so rapidly, its ability to act as a stimulant is weak and short-acting.3
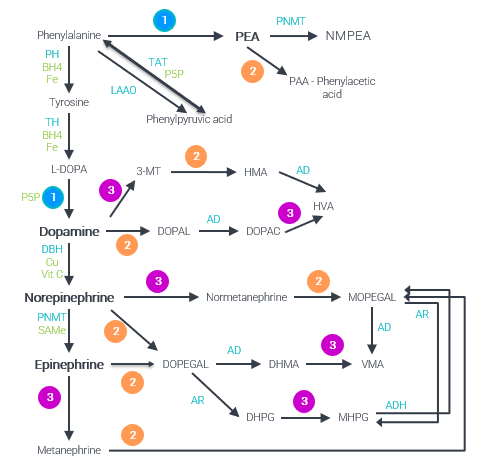
Phenylethylamine Synthesis. PEA is produced from phenylalanine and requires vitamin B6. It is rapidly degraded to phenylacetic acid and N-Methylphenethylamine (NMPEA). Vitamin B2 is a cofactor in the breakdown of PEA to phenylacetic acid (PAA).
Enzymes and Cofactors found in Multiple Monoamine Pathways 1) Aromatic L-amino acid decarboxylase (AADC) + Vitamin B6 2) Monoamine oxidase (MAO) + Vitamin B2 3) Catechol-O-Methyltransferase (COMT) + Magnesium + S-adenosylmethionine (SAMe) Neurotransmitters and Metabolites DHPG: 3,4-dihydroxyphenylglycol, DHMA: 3,4-dihydroxymandelic acid, DOPAC: 3,4-dihydroxyphenylacetic acid, DOPAL: 3,4-dihydroxyphenylacetaldehyde, DOPEGAL: 3,4-dihydroxyphenylglycolaldehyde, HMA: 4-hydroxy-3-methoxyphenylacetaldehyde / homovanillin, HVA: homovanillic acid, MHPG: 3-methoxy-4-hydroxyphenylglycol (aka MOPEG), MOPEGAL: 3-methoxy-4-hydroxyphenylglycolaldehyde, NMPEA: N-Methylphenethylamine, VMA: vanillylmandelic acid, 3-MT: 3-methoxytyramine, Enzymes AD: aldehyde dehydrogenase, ADH: alcohol dehydrogenase, AR: aldehyde reductase, DBH: Dopamine Beta-Hydroxylase, LAAO: L-amino-acid oxidase, PH: alanine Hydroxylase, PNMT: phenylethanolamine N-methyltransferase, TAT: Tyrosine aminotransferase, TH: Tyrosine hydroxylase, Cofactors BH4: Tetrahydrobiopterin, Fe: Iron, SAMe: S-adenosylmethionine, Vit: Vitamin
What Does PEA Do in The Body?
PEA is considered an endogenous amphetamine, but its stimulatory abilities are weak and short.5 PEA levels made by the body under normal physiological conditions support healthy energy levels, which is why it is often of interest to athletes.
PEA As A Nootropic
PEA is a nootropic, a substance that supports mental performance. Through its ability to amplify the effects of other neurotransmitters and brain chemicals including acetylcholine,6 PEA increases motivation for finishing tasks and enhances cognitive function and problem-solving abilities. Acetylcholine acts as a neurotransmitter and neuromodulator that plays a role in many bodily functions, particularly learning and memory. By enhancing acetylcholine actions, PEA can support cognitive function and improve focus and attention span.7
Enhances Mood
Balanced levels of PEA contribute to happiness, pleasure, emotional well-being, and having an upbeat attitude.4 Levels of the PEA metabolite, PAA, are low in people who are unhappy.8 Conversely, employing strategies to improve mood in this group of people raises PEA concentrations in the brain. PAA itself acts like a natural endorphin, producing what’s known as a “runner’s high.”4
What Are The Effects of Low PEA and High PEA?
Low Phenylethylamine is linked to:1,7,8
- Brain fog
- Cravings
- Low energy levels
- Inability to concentrate
- Not feeling motivated
- Social introversion
- Unhappiness
High PEA may be associated with:
- Jitteriness
- Increased heart rate
- Muscle stiffness
- Nervousness
- Shakiness
- Rising blood pressure
What Are the Causes of Phenylethylamine Deficiency?
Alterations in PEA levels may occur due to changes in AADC or MAO-B, the enzymes involved in PEA synthesis and breakdown. Low dietary protein, maldigestion, or malabsorption can impact PEA levels, as protein is a source of L-phenylalanine. Lack of exercise is another contributor to low PEA levels (more on this later).
How Does PEA Interact with Other Neurotransmitters?
PEA boosts the effectiveness and actions of catecholamine neurotransmitters. Most evidence indicates it both amplifies the release of and competitively blocks the reuptake of all the monoamine : dopamine, norepinephrine, and serotonin. PEA is localized around dopaminergic neurons and regulates dopaminergic neurotransmission.10 It plays a role in dopamine’s pleasure-producing effects. High doses of PEA significantly increase serotonin levels, but serotonin can inhibit PEA.11,12
PEA Testing
Urinary neurotransmitter testing for PEA has been used as a screening tool for monitoring PEA levels in various populations, including individuals seeking to support attention and focus, learning and memory, and mood. Major universities and research facilities throughout the world continue to use this technology for measuring neurotransmitter levels in patients with a variety of mental health conditions.
PEA testing using NeuroLab® is an excellent clinical tool for diagnosing PEA deficiency and excess as well as imbalances in other neurotransmitters. Utilizing the gold standard in urinary neurotransmitter testing, NeuroLab® employs UHPLC Triple Quadrupole Mass Spectrometry (MS) technology in order to provide the highest level of sensitivity and specificity for testing neurotransmitters. PEA urinary testing is a convenient way for patients to test at home without the need for a blood draw.
How Can We Naturally Support PEA Levels?
Several strategies can be used to support healthy levels of PEA.
PEA as a Nootropics Supplement
Phenylethylamine can be consumed as a dietary supplement. The typical phenylethylamine dose is 100 to 500 mg daily. Clinicians often start their patients on a lower dose and gradually increase the amount. PEA should also not be taken together with medications that increase serotonin levels as this can cause an excess in serotonin. Testing PEA and serotonin levels before beginning supplementation is recommended.
Which Foods Contain PEA?
PEA is found in a number of food sources. 1 It’s produced through bacterial fermentation, especially Bacillus subtilis, so some fermented foods can be an excellent source of PEA.13
- Almonds
- Blue green algae
- Chickpeas
- Chocolate
- Eggs
- Flaxseeds
- Green peas
- Lentils
- Natto and some cheeses
- Soybeans
- Walnuts
- Wine
PEA is also made through the conversion of L-phenylalanine from food. Consequently, eating high-protein foods rich in L-phenylalanine such as eggs, chicken, turkey, beef, and dairy, can also raise PEA levels.
Supplementation of L-phenylalanine, vitamin B6, and vitamin B2, riboflavin can improve PEA synthesis and clearance.
Exercise and PEA
Exercise is known to elevate levels of PEA and/or its metabolites. For example, in one study, the PEA metabolite PAA increased 77% after exercise, according to 24-hour urine samples from healthy men.14 The researchers concluded that exercise likely increases PEA, which could explain the known mood-boosting effect of physical activity.
References
- ScienceDirect. Published 2009. https://www.sciencedirect.com/topics/neuroscience/trace-amine.
- Pei Y, Asif-Malik A, et al. Front Neurosci. 2016;10:148.
- Janssen PA, Leysen JE, et al. Int J Neuropsychopharmacol. 1999;2(3):229-240.
- Irsfeld M, Spadafore M, et al. Webmedcentral. 2013;4(9).
- Berry MD. J Neurochem. 2004;90(2):257-271.
- Kato M, Ishida K, et al. Eur J Pharmacol. 2001;418(1-2):65-71.
- Ishida K, Murata M, Kato M, et al. Biol Pharm Bull. 2005 Sep;28(9):1626-9.
- Sandler M, Ruthven CR, et al. Clin Chim Acta. 1979;93(1):169-171.
- Sotnikova TD, Budygin EA, et al. J Neurochem. 2004;91(2):362-373.
- Nakamura M, Ishii A, et al. Eur J Pharmacol. 1998;349(2-3):163-169.
- Goudie AJ, Buckland C. Neuropharmacology. 1982;21(12):1267-1272.
- Kim B, Byun BY, et al. Food Chem. 2012;135(3):2005-2011.
- Szabo A, Billett E, et al. Br J Sports Med. 2001;35(5):342-343.