SAD, or seasonal affective disorder, is a disorder in which there may be both dysfunction of the circadian rhythm and production of excess melatonin during the long nights of winter. Symptomology includes profound depression, hypersomnia, carbohydrate cravings/weight gain, fatigue, sadness and often a reduction of socialization. It is estimated that 10 to 20 percent of recurrent depression cases follow a seasonal pattern.[1] SAD is a relatively common condition and has been found to be up to four times more prevalent in women.[2],[3] In U.S. community surveys, SAD prevalence ranges from 9.7 percent in New Hampshire to 1.4 percent in Florida.[4] In North America, SAD prevalence increases significantly with latitude. In Europe, oddly enough, the correlation between seasonal affective disorder with latitude was found nonsignificant, only trending in that direction.[5]
Seasonal affective disorder has been regarded primarily as a melatonin disorder. For SAD patients, the nocturnal period of active melatonin secretion is longer in winter than in summer. In healthy volunteers, however, there is no change.[6] Karadottir and Axelsson found that SAD patients’ saliva, collected in March of the study year, had 2.4 times more melatonin than controls.[7] This change in melatonin levels is similar to the signal that other mammals use to regulate seasonal changes in their behavior. However, the pathophysiological mechanisms of SAD may be more complex than just melatonin. Hypotheses on other underlying mechanisms indicate that environmental variables, e.g. climate, latitude, light, circadian rhythm, and changes in neurotransmitter function that occur naturally with the seasons, may also be important.[8]
Neurotransmitter Involvement in Seasonal Affective Disorder
Neurotransmitter studies support a role for serotonin, norepinephrine and dopamine in the affective and/or appetitive symptoms of SAD.
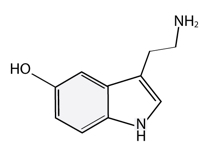
Of the three, serotonin is by far the most well-studied. Serotonin has the clearest seasonal rhythm in its metabolism and availability, with most measures pointing to decreased levels/activity in the winter months.[9] For example, serotonin was found to be higher in summer and after light treatment (discussed below) than in winter in both SAD patients and controls.[10] Tyrer and colleagues recently found that serotonin transporter (SERT) binding potential was increased in SAD, and the higher the increase, the worse the symptomology.[11] McMahon et al, summed up with the explanation that depressive symptoms in winter are associated with a failure to downregulate serotonin transporter levels appropriately during exposure to the environmental stress of winter.[12] That is, SERT is in a hyperfunctional state during SAD; it removes too much serotonin from the synapse, decreasing serotonin activity during the short photoperiod of winter.[13] Certain serotonergic gene polymorphisms have also been connected to SAD. Sher reports the 5-HTTLPR and the 5-HT(2A)-1438G/A gene promoter polymorphisms are associated with SAD.[14] So we see a genetic influence on serotonin levels, increasing SAD risk as well.
Increasing serotonin has shown consistent efficacy in treating SAD. For example, increasing serotonin raised SAD depression into euphoria and blunted HPA and SNS responses in untreated SAD patients but not treated ones.[15] SSRIs and to some extent, SNRIs have been successfully used to treat SAD symptoms, as well.[16]
Tryptophan depletion studies also support serotonergic involvement in winter depression. Lam, et al. found that rapid tryptophan depletion reversed the antidepressant effect of light therapy in SAD patients.[17] Supplementing tryptophan has been shown to be effective in the treatment of SAD patients resistant to light therapy as well as in prolonging the antidepressant effects of light therapy.[18] Another aspect of serotonin’s involvement involves carbohydrate cravings. Sugar-rich diets can attenuate the carb cravings that occur when serotonin falls.[19] This is because carbohydrate-rich diets trigger an insulin response which enhances the bioavailability of tryptophan in the CNS. As tryptophan crosses the blood-brain barrier more easily under the influence of insulin, serotonin production rises, reducing the craving of carbohydrates.[20] Rosenthal et al. studied carbohydrate cravings in SAD patients. SAD patients reported activation following carbohydrate ingestion, whereas normal controls reported sedation.[21] We can see that, for SAD patients, low serotonin, tryptophan availability and transport and an altered response to carbohydrate all coalesce to produce a part of SAD symptomology.
Norepinephrine and dopamine may also be involved. Catecholamine depletion in SAD patients during the summer months causes SAD relapse.[22] Also, one study found an increase in plasma levels of norepinephrine following light treatment for SAD.[23] Reboxetine, an SNRI that blocks norepinephrine reuptake, was equally as effective as the SSRI, escitalopram in relieving SAD symptoms.[24] It is apparent that norepinephrine plays a role and may need to be optimized when addressing SAD.
Dopamine is implicated as well. According to Levitan’s research on SAD, the Dopamine 4 receptor gene may be involved, in that the “hypofunctional 7-repeat allele of DRD4” has been linked to both affective and appetitive symptoms in SAD.[25]
There is also a hypothesis that vitamin D status is important. Levels do fluctuate seasonally and low D status has been observed in depression.[26] No studies have been reported, however, on vitamin D and SAD, but optimization of D levels likely should be on the therapeutic protocol for SAD.
Light Therapy
The most effective treatment for SAD, however, is light therapy. The antidepressant response is contingent on the exposure of the patients’ eyes to light in the range of 2000 to 10,000 lux. In classical light therapy, the patient sits in front of the light in the morning for 30 to 120 minutes daily during the winter.[27] Narrow bandwidth blue light has been shown more efficacious than red light.[28] However, Anderson and colleagues recently found white light to be as successful as blue light for SAD.[29] Studies report effectiveness from between 53% to 76%.[30],[31]
The mechanism of action of light therapy is not fully known. The phase shift hypothesis states that most patients with SAD become depressed in the winter because of a delay in circadian rhythms with respect to the sleep/wake cycle. It is reported that many, but not all SAD patients are phase-delayed and thus light treatment works to return their circadian rhythm to normal.[32] We do know that light treatment (as well as a change in the season) creates a phase advance shift in melatonin rhythm (i.e., a patient goes to sleep earlier).[33] Serotonin dysfunction may also play a role in the success of light therapy. Neumeister et al. found that tryptophan depletion caused a relapse in remission in patients undergoing light therapy, concluding that light therapy needed a functional serotonin system to maintain remission.[34] Tyrer et al. found that light therapy reduced SERT binding potential, also supporting the serotonin connection to light’s efficacy.[35]
Much research has been done on seasonal affective disorder and certain interventions have remarkable success in attenuating symptoms. Including neurotransmitter assessment is useful to be able to pinpoint which neurotransmitter(s) are imbalanced. Combining neurotransmitter optimization with light therapy often goes a long way in producing a bright, sunny smile in the middle of winter.
References
[1] Roecklein KA, Rohan KJ. (2005). Seasonal affective disorder: an overview and update. Psychiatry (Edgmont). Jan;2(1):20-6.
[2] Magnusson A, Boivin D. (2003). Seasonal affective disorder: an overview. Chronobiol Int. Mar;20(2):189-207.
[3] Gupta A, Sharma PK, Garg VK, et al. (2013). Role of serotonin in seasonal affective disorder. Eur Rev Med Pharmacol Sci. Jan;17(1):49-55.
[4] Roecklein KA, op. cit.
[5] Mersch PP, Middendorp HM, Bouhuys AL, et al. (1999). Seasonal affective disorder and latitude: a review of the literature. J Affect Disord. Apr;53(1):35-48.
[6] Wehr TA, Duncan WC Jr, Sher L, et al. (2001). A circadian signal of change of season in patients with seasonal affective disorder. Arch Gen Psychiatry. Dec;58(12):1108-14.
[7] Karadottir R, Axelsson J. (2001). Melatonin secretion in SAD patients and healthy subjects matched with respect to age and sex. Int J Circumpolar Health. Nov;60(4):548-51.
[8] Attar-Levy D. (1998). [Seasonal depression]. Therapie. Sep-Oct;53(5):489-98.
[9] Levitan RD. (2007). The chronobiology and neurobiology of winter seasonal affective disorder. Dialogues Clin Neurosci. 9(3):315-24.
[10] Danilenko KV, Putilov AA, Russkikh GS, et al. (1994). Diurnal and seasonal variations of melatonin and serotonin in women with seasonal affective disorder. Arctic Med Res. Jul;53(3):137-45.
[11] Tyrer AE, Levitan RD, Houle S, et al. (2016). Increased Seasonal Variation in Serotonin Transporter Binding in Seasonal Affective Disorder. Neuropsychopharmacology. Sep;41(10):2447-54.
[12] Mc Mahon B, Andersen SB, Madsen MK, et al. (2016). Seasonal difference in brain serotonin transporter binding predicts symptom severity in patients with seasonal affective disorder. Brain. May;139(Pt 5):1605-14.
[13] Willeit M, Sitte HH, Thierry N, et al. (2008). Enhanced serotonin transporter function during depression in seasonal affective disorder. Neuropsychopharmacology. Jun;33(7):1503-13.
[14] Sher L. (2001). Genetic studies of seasonal affective disorder and seasonality. Compr Psychiatry. Mar-Apr;42(2):105-10.
[15] Schwartz PJ, Murphy DL, Wehr TA, et al. (1997). Effects of meta-chlorophenylpiperazine infusions in patients with seasonal affective disorder and healthy control subjects. Diurnal responses and nocturnal regulatory mechanisms. Arch Gen Psychiatry. Apr;54(4):375-85.
[16] Michalak EE, Murray G, Levitt AJ, et al. (2007). Quality of life as an outcome indicator in patients with seasonal affective disorder: results from the Can-SAD study. Psychol Med. May;37(5):727-36.
[17] Lam RW, Zis AP, Grewal A, et al. (1996). Effects of rapid tryptophan depletion in patients with seasonal affective disorder in remission after light therapy. Arch Gen Psychiatry. 1996 Jan;53(1):41-4.
[18] Kulikov AV, Popova NK. (2015). Tryptophan hydroxylase 2 in seasonal affective disorder: underestimated perspectives? Rev Neurosci. 26(6):679-90.
[19] Inam QU, Ikram H, Shireen E, et al. (2016). Effects of sugar rich diet on brain serotonin, hyperphagia and anxiety in animal model of both genders. Pak J Pharm Sci. May;29(3):757-63.
[20] Shabbir F, Patel A, Mattison C, et al. (2013). Effect of diet on serotonergic neurotransmission in depression. Neurochem Int. Feb;62(3):324-9.
[21] Rosenthal NE, Genhart MJ, Caballero B, et al. (1989). Psychobiological effects of carbohydrate- and protein-rich meals in patients with seasonal affective disorder and normal controls. Biol Psychiatry. Apr 15;25(8):1029-40.
[22] Lam RW, Tam EM, Grewal A, et al. (2001). Effects of alpha-methyl-para-tyrosine-induced catecholamine depletion in patients with seasonal affective disorder in summer remission. Neuropsychopharmacology. Nov;25(5 Suppl):S97-101.
[23] Anderson JL, Vasile RG, Mooney JJ, et al. (1992). Changes in norepinephrine output following light therapy for fall/winter seasonal depression. Biol Psychiatry. Oct 15;32(8):700-4.
[24] Pjrek E, Konstantinidis A, Assem-Hilger E, et al. (2009). Therapeutic effects of escitalopram and reboxetine in seasonal affective disorder: a pooled analysis. J Psychiatr Res. May;43(8):792-7.
[25] Levitan, op. cit.
[26] Stewart AE, Roecklein KA, Tanner S, et al. (2014). Possible contributions of skin pigmentation and vitamin D in a polyfactorial model of seasonal affective disorder. Med Hypotheses. Nov;83(5):517-25.
[27] Magnusson, op. cit.
[28] Glickman G, Byrne B, Pineda C, et al. (2006). Light therapy for seasonal affective disorder with blue narrow-band light-emitting diodes (LEDs). Biol Psychiatry. Mar 15;59(6):502-7.
[29] Anderson JL, Hilaire MA, Auger RR, et al. (2016). Are short (blue) wavelengths necessary for light treatment of seasonal affective disorder? Chronobiol Int. Aug 5:1-13.
[30] Goel N, Terman M, Terman JS, et al. (2005). Controlled trial of bright light and negative air ions for chronic depression. Psychol Med. Jul;35(7):945-55.
[31] Attar-Levy, op. cit.
[32] Lewy AJ, Emens JS, Songer JB, et al. (2009). Winter Depression: Integrating mood, circadian rhythms, and the sleep/wake and light/dark cycles into a bio-psycho-social-environmental model. Sleep Med Clin. Jun 1;4(2):285-299.
[33] Danilenko, op. cit.
[34] Neumeister A, Praschak-Rieder N, Besselmann B, et al. (1997). Effects of tryptophan depletion on drug-free patients with seasonal affective disorder during a stable response to bright light therapy. Arch Gen Psychiatry. Feb;54(2):133-8.
[35] Tyrer AE, Levitan RD, Houle S, et al. (2016). Serotonin transporter binding is reduced in seasonal affective disorder following light therapy. Acta Psychiatr Scand. Nov;134(5):410-419.